The compound played an important role in the development of sandwich compounds in organometallic chemistry and is the prototypical complex containing two arene ligands. 
It is sometimes called dibenzenechromium. Also replace URL for the actual url of this page (The stay, ok?). Bis (benzene)chromium is the organometallic compound with the formula Cr ( 6 -C 6 H 6) 2. Now replace dd, mmmm and yyyy with the day, month, and year you browsed this page. Total valence electrons given by chromium atom 61 6. "Valence Electrons in Chromium (Cr) [& Facts, Color, Discovery. So, both chromium and oxygen atoms have six electrons in their valence shells. To make your life (and citation) easier just copy & paste the information below into your assignment or essay: That gives credibility to your paper and it is sometimes required in higher education. CitationWhen you need to include a fact or piece of information in an assignment or essay you should also include where and how you found that piece of information. How about an incentive to share this post? (You will help other colleagues find this blog)ĭownload and enjoy this complete and colored periodic table for you to edit and enjoy. 
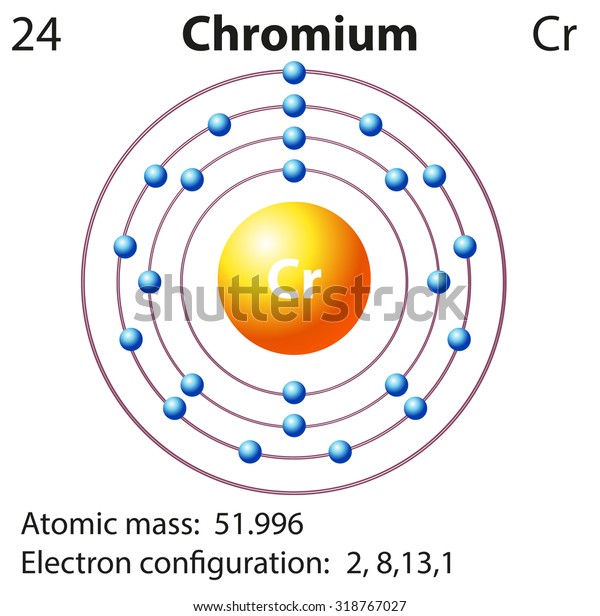
Need an editable periodic table to edit? Maybe add your school logo, work team or anything else to maker your paper look cool?Īlong with basic atom / element information (like Chromium valence electrons and all the other atomic data), it also comes with color coded info about: State (Gas, Liquid or Solid at room temperature), Groups/series details and much more. How a small number of atoms can be joined and form completely different substances.Electron affinity increases from bottom to top in a family of nonmetal elements on the periodic table. Chromium has lower ionization energy than zinc. Video Are you having trouble understanding the basics of atomic elements? This video will walk you through: Chromium has more valence electrons than zinc. Want to learn more details and data about Chromium (Cr)? Check my Elements Comprehensive List. Produced commercially by heating its ore in the presence of silicon or aluminium. SourcesChromite is its most important mineral. Iron-nickel-chromium alloys in various percentages yield an incredible variety of the most important metals in modern technology. It gives the color to rubies and emeralds. It is hard, brittle and corrsion-resistant at normal temperatures. In the case of Chromium the valence electrons is 2,3,6. Now let's check the facts about Chromium.ĭescriptionVery hard, crystalline, steel-gray metal. Ok but how many valence electrons does an atom of Chromium have? A valence electron is an outer shell electron and may participate in the formation of a chemical bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
